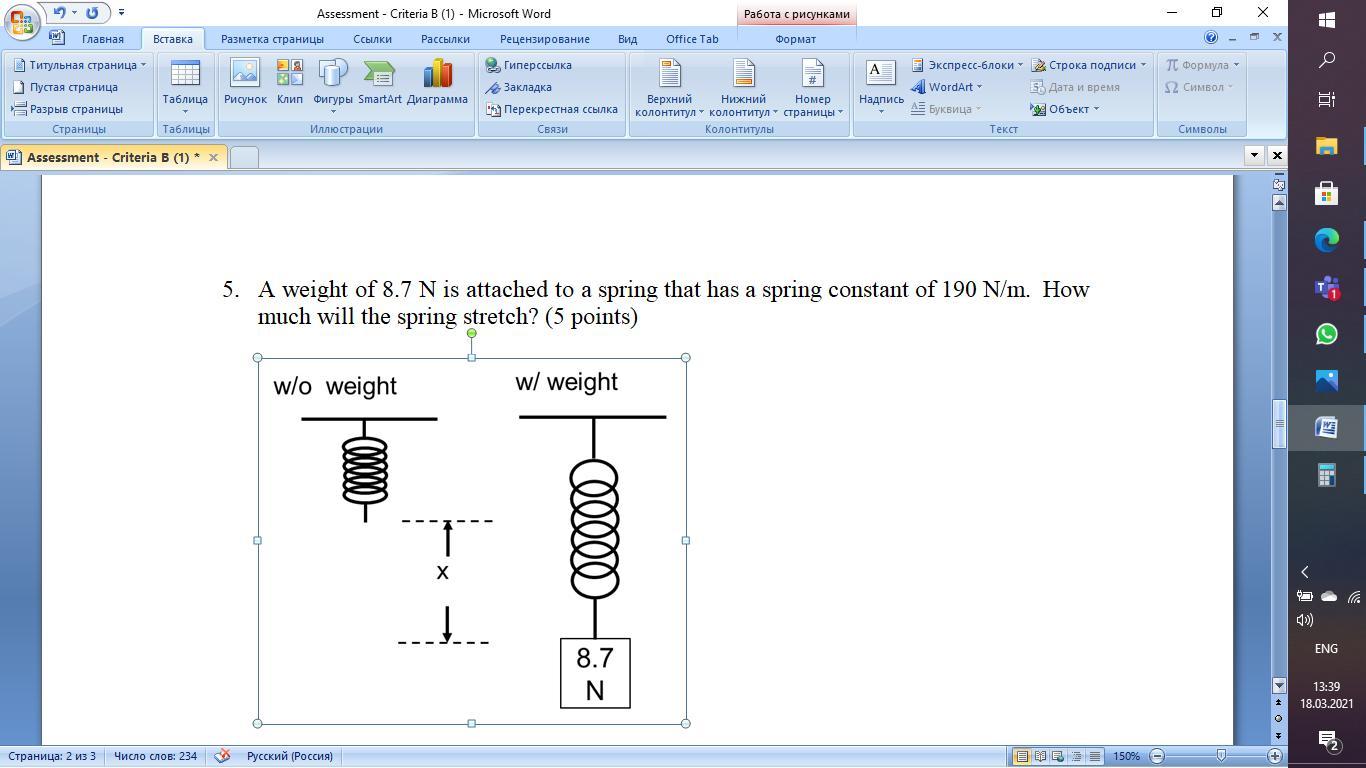
A weight of 8.7 N is attached to a spring that has a spring constant of 190 N/m. How much will the spring stretch?
Answers
Fs=8.7N
K=190N/m
Unkown:
X=?
Equation:
Fs=Kx
8.7N=(190N/m)x
X=4.6*10power of-2m
Given values are:
Force, Fs = 8.7 NSpring constant, k = 190 N/mAs we know the relation,
→ [tex]Fs = K\times x[/tex]
or,
→ [tex]x = \frac{Fs}{k}[/tex]
By putting the values, we get
[tex]= \frac{8.7}{190}[/tex]
[tex]= 4.6\times 10^{-2} \ m[/tex]
Thus the response above is right.
Learn more about spring constant here:
https://brainly.com/question/15404289
Related Questions
Under each photo,
write an example of where you
might find water in that state of
matter.
Answers
Answer:
Explanation:
water vapor:in a cloud or when you breath out once it's cold.Ice: in your freezer or on a frozen lake.liquid water:at a stream
what physical properties does lithium have
Answers
Answer:
Lithium has a melting point of 180.54 C, a boiling point of 1342 C, a specific gravity of 0.534 (20 C), and a valence of 1. It is the lightest of the metals, with a density approximately half that of water. Under ordinary conditions, lithium is the least dense of the solid elements.
Appearance: soft, silvery-white metal
Atomic Number: 3
Atomic Radius (pm): 155
How many molecules are in 41.8 g H2O?
Answers
Answer:
7.63 × 10²³ molecules
Explanation:
First, convert grams to moles using the molar mass of water (32.988 g/mol).
41.8 g ÷ 32.988 g/mol = 1.267 mol
Next, convert moles to molecules using Avogadro's number (6.022 × 10²³).
1.267 mol × 6.022 × 10²³ molecules/mol = 7.63 × 10²³ molecules
Why is chemistry important? i need 2 paragraph please
Answers
Plants and animals require large amounts of water in order to survive. Although vast numbers of plants and animals have lived throughout time, the amount of water in our environment has remained relatively constant. Which of the following statements explains why this occurs?
A. Water continually flows from the center of Earth.
B. Vast quantities of water are created by lightning in clouds.
C. Organisms return water to the environment after they use it.
D. Organisms combine hydrogen and oxygen to make their own water.
please help me
Answers
Answer: C - Organisms return water to the environment after they use it.
Explanation: The amount of water on earth will always remain the same. Water goes in and out of all organisms. It is not possible to be more or less.
My feet get hot as soon as I touch the sand. What kind of heat transfer occurred?
Answers
please mark brainliest
Please help! Find the distance to each destination in AU. Use the correct conversion factor.
Answers
Explanation:
The question involves converting from one unit (Kilometres) to another unit (Astronomical unit)
The relationship between both units is given as;
1 Km = 6.68459e-9 Au
Kulper belt
6.5 billion km = 6.5e9 Km
1 Km = 6.68459e-9 Au
6.5e9 Km = x
Solving for x;
x = 43.45 Au
Alpha Centauri
44 trillion km = 4.4e13
1 Km = 6.68459e-9 Au
4.4e13 km = x
Solving for x;
x = 294121.83 Au
Cart A has a mass of 24 kg and is being pulled with a force of 32 N. Cart B has a mass of
27 kg and is being pulled with a force of 33 N. Which cart will have the faster acceleration?
Answers
Please help me on this.
Answers
Answer: true
Explanation:
i hope this helps
How many moles are in 281 g of Ca(OH)2?
Answers
Answer:
3.79 moles
Explanation:
To convert moles to gams of a substance we need to find the molar mass of the substance. For Ca(OH)₂ th molar mass is:
1Ca = 40.08g/mol
2O = 2*16g/mol = 32g/mol
2H = 2*1.01g/mol = 2.02g/mol
The molar mass is:
40.08g/mol + 32g/mol + 2.02g/mol = 74.1g/mol
And moles are:
281g * (1mol / 74.1g) =
3.79 molesWhich of the following factors would affect the rate at which the stages of death happen
A.Humidity
B.Temperature
C.If the person was sick
D.All of the choice would affect the rate
Answers
Does sanitizer conduct electricity
Answers
Answer:
Yes, since it has alcohol, it conducts electricity.
Another alloy called nichrome contains only the elements chromium and nickel how much nickel dies it have
Answers
Answer:
80 g
Explanation:
Alloy = nichrome
100 g of nichrome contains 20 g of chromium
Contains only two elements (Nickel and Chromium) hence;
Mass of Alloy = Mass of Nickel + Mass of Chromium
100 = Nickel + 20g
Nickel = 100 g - 20 g = 80 g
calculate the [H+] if the [OH-] is 7.34x10^-10
pls show work
Answers
7. H3PO4 →
H4P207+
H20
Balance the equation
Answers
This should hopefully help
2H³PO⁴ --> H⁴P²O⁷ + H²O
3. What tool do we use to measure force?
Answers
Answer:
A force meter
The chemical formula of aspirin is C9H8O4. What is the mass of 0.40 mol
of aspirin?
Answers
Answer:
[tex]\boxed {\boxed {\sf About \ 72 \ grams }}[/tex]
Explanation:
To convert from moles to grams, we must use the molar mass (also known as the gram formula mass).
First, look up the molar masses of the elements in the formula.
C: 12.011 g/mol H: 1.008 g/mol O: 15.999 g/molNext, multiply by the subscript, because it tells us the number of atoms of each element in the formula.
C₉: 4(12.011 g/mol)= 108.099 g/mol H₈: 8(1.008 g/mol)= 8.064 g/mol O₄: 4(15.999 g/mol)= 63.996 g/molAdd the values.
108.099 + 8.064+63.996=180.159 g/molUse this molar mass as a ratio.
[tex]\frac {180.159 \ g \ C_9H_8O_4}{1 \ mol \ C_9H_8O_4}[/tex]
Multiply by the given number of moles, 0.40
[tex]0.40 \ mol \ C_9H_8O_4 *\frac {180.159 \ g \ C_9H_8O_4}{1 \ mol \ C_9H_8O_4}[/tex]
The units moles of aspirin cancel.
[tex]0.40 *\frac {180.159 \ g \ C_9H_8O_4}{1 }[/tex]
[tex]72.0636 \ g \ C_9H_8O_4[/tex]
The original number of moles has 2 sig figs (4 and 0), so answer must have the same. For the number we calculated, it is the ones place. The 0 in the tenth place tells us to leave the 2.
[tex]72 \ g[/tex]
Identify the shape below:
PLEASE HELP
Answers
Answer:
linear angleee
Explanation:
lol i dont know what to put here
Answer:
B) Bent
There are a lot of types of bents but this one is linear bent.
PLEASE HELP: 60 POINTS
Select all that apply!
The emission of a Beta particle has the effect of:
a change in mass of an electron emitted from the nucleus
decreasing mass by one unit
increasing mass by one unit
adding a proton to the nucleus
converting 1 neutron to 1 electron and 1 proton
Answers
Answer:
all of they above
Explanation:
specific heat is defined as the amount of heat in what
Answers
Answer:
Specific heat is the amount of energy that must be added, in the form of heat, to one unit of mass of the substance in order to cause an increase of one unit in temperature.
Explanation:
Answer:
Specific heat is the amount of energy that must be added, in the form of heat, to one unit of mass of the substance in order to cause an increase of one unit in temperature.
Explanation:
CAN SOMEONE PLEASE HELP ME WITH THIS?!!!!
Answers
Answer:
1. A. 9 eggs
2. B. 5:3
3. C. Exactly 44 g
4. C. Mass and atoms only
5. D. 6.81 g of PH₃
6. C. 88.4%
7. B. O₂
8. D. 30.8 g CO₂
9. B.
10. B.
11. C. 99 g
12. D. mole ratio
13. C. Theoretical
14. A. 6.0 mol H₂O
15. D. 39.7 g CH₃OH
16. A. 650 g HgO
17. D. 8.8 mol H₂
18. B. 82.6%
Explanation:
1. The number of eggs it takes to make 1 cake = 3 eggs
The number of eggs it takes to make 3 × 1 = 3 cake = 3 × 3 = 9 eggs
Therefore, the correct option is;
A. 9 eggs
2. The given reaction is presented as follows;
C₃H₈(g) + 5O₂(g) → 3CO₂(g) + 4H₂O(l)
In the above reaction, we have;
Moles of , O₂, reacted = 5 moles
Moles of , CO₂, produced = 3 moles
The ratio of the of O₂ reacted to moles of CO₂ produced = 5 moles:3 moles
∴ The ratio of the of O₂ reacted to moles of CO₂ produced = 5:3
The correct option is;
B. 5:3
3. The reaction is presented as follows;
O₂ (g) + C(s) → CO₂ (g)
From the reaction, 1 mole (12 g) of carbon produces 1 mole of CO₂
The molar mass of CO₂ = The mass of 1 mole of CO₂ = 44.01 g/mol
Given that the reaction is completed, the mass of CO₂ produced = The mass of 1 mole of CO₂ ≈ 44 g
The correct option is;
C. Exactly 44 g
4. The given reaction is presented as follows;
N₂ + 3 F₂ → 2NF₃
The initial number of atom = 2 + 6 = 8
The final number of atom = 2 × 4 = 8
∴ The initial number of atom = The final number of atom
Therefore, the number of atoms is conserved;
The mass of the reactants ≈ 28 g/mol + 3 × 37.996806 g/mol ≈ 141.993612 g/mol ≈ 142 g/mol
The mass of the product ≈ 2 × 71 g.mol = 142 g/mol
∴ The mass is conserved
Moles of reactants = 1 + 3 = 4
Moles of products = 2
∴ The number of moles is not conserved
The correct option is
C. Mass and atoms only
5. The molar mass of P₄ = 123.895048 g/mol
One mole of P₄ (123.895048 g) produces four moles (4 × 34.00) of PH₃
6.20 g of P₄. will produce (4 × 34.00)/(123.895048) × 6.20 g ≈ 6.80576 g ≈ 6.81 g
The correct option is D. 6.81 g of PH₃
6. The percentage yield = ((The actual yield)/(The ideal yield)) × 100
The actual yield of silver = 38.1 g
The ideal yield of silver = 43.1 g
∴ The percentage yield = ((38.1 g)/(43.1 g)) × 100 = 88.3990719258% ≈ 88.4%
The percentage yield = 88.4%
The correct option is C. 88.4%
7. The given chemical equation is presented as follows;
CS₂ (g) + 3 O₂ (g) → CO₂ (g) + 2 SO₂ (g)
The number of moles in 192 g of O₂ = 192 g/(32 g/mol) = 6 moles
Given that 3 moles of O₂ reacts with 1 mole of CS₂ to produce 1 mole of CO₂ and 2 moles SO₂, therefore 2 × 3 = 6 moles of O₂ will reacts with 2 × 1 = 2 moles of CS₂ to produce 2 moles of CO₂ and 4 moles SO₂
∴ The limiting reactant is;
B. O₂
8. The given chemical equation is presented as follows;
2 C₈H₁₈ (g) + 25 O₂ (g) → 16 CO₂ (g) + 18 H₂O (l)
The number of moles in 10 g of C₈H₁₈, n₁ = (10 g)/(114.26 g/mol)
The number of moles of CO₂ produced, n₂ = (10 g)/(114.26 g/mol) × 16/2 ≈ 0.7 moles
The mass of CO₂ produced, m ≈ 44.01 × n₂ ≈ 44.01 g/mol × 0.7 moles ≈ 30.807 grams ≈ 30.8 grams
The theoretical yield of CO₂ from completely burning 10.0 g of C₈H₁₈ ≈ 30.8 grams of CO₂
The correct option is D. 30.8 g CO₂
9. The correct option is B. The limiting reactants determine the maximum amount of product that can be formed
10. Option B, which has 3 atoms of each element combining to form a product with 1 atom of one element and 2 atoms of the other element
The correct option is B.
11. By the conservation of mass, we have;
The mass of the reactants = The mass of the products
Let 'x' represent the mass of zinc in the products of the reaction
Therefore, we have;
61 g of calcium + 207 g of zinc chloride = 169 g of calcium chloride + x g of Zinc
∴ x g = 61 g + 207 g - 169 g = 99 g
The mass of zinc in the products of the reaction, x g = 99 g
The correct option is;
C. 99 g
12. The quantity that must be used to convert from one chemical substance to another is the mole ratio
The correct option is D. mole ratio
13. The maximum mass of the product that could form in a reaction is called the theoretical yield, which is option C.
The correct option is C. Theoretical
14. 1 mole of O₂ produces 2 moles of water (H₂O), therefore;
3 × 1 = 3.0 moles of O₂ will produce 3 × 2 = 6 moles of H₂O
The correct option is
A. 6.0 mol H₂O
15. 2 mole × 2.02 g/mol = 4.04 g of H₂ (g) produces 32.05 g CH₃OH (l)
∴ 5 g of H₂ (g) will produce 32.05 g × 5/4.04 ≈ 39.6658416 g ≈ 39.7 g of CH₃OH
The correct option is;
D. 39.7 g CH₃OH
16. 2 (2 × 216.59 g = 433.18 g) moles of HgO produces 1 mole of O₂
1.5 mole of O₂ will be produced by 1.5 × 2 = 3 moles (3 × 216.59 g = 649.77 g ≈ 650 g) of HgO
The correct option is A 650 g HgO
17. 3 moles of H₂ produces 2 moles of NH₃
The number of moles of NH₃ in 100 g of NH₃, n = 100 g/(17.04 g/mol) = 5.868544 moles
The number of moles of H₂ that will produce 5.868544 moles of NH₃ = 3/2 × 5.868544 moles = 8.802816 moles ≈ 8.8 moles
Therefore, the correct option is;
D. 8.8 mol H₂
18. The theoretical yield of PbO = (223.2/331.2) × 9.90 g = 6.67173913 g
The percentage yield = (5.51 g)/(6.67173913 g) × 100 ≈ 82.6%
The correct option is option B 82.6%.
Elements in a convolent bond are called ?
Answers
Answer:
A covalent bond is formed by an equal sharing of electrons from both the participating atoms. The pair of electrons participating in this type of bonding is called shared pair or bonding pair. The covalent bonds are also termed molecular bonds.
Can someone please help I need help?!?!
Answers
Answer:
a: 6 moles
b: 6.75 moles
c: 5 grams
Explanation:
a: mole ratio 2:3
b: mole ratio 2:3
c: mole ratio 2:2
Why is it not possible to extract aluminium by heating its ore with carbon?
Answers
If I touch a hot object, my hand gets hot through which type of heat transfer?
Answers
Answer:
Thermal Transfer.
Explanation:
If you touch a hot object, the heat is transferred from the object to your hand through conduction. (This is the actual answer.)
If the mass of all the reactants in a chemical reaction is 100g, what will the mass of all products be?
Answers
K(s)+H2SO4(aq)
——>_____+______
Answers
K2SO4 + H2
Reason :Potassium is highly reactive
Answer:
K2SO4 + H2 this is the reaction.
2 HCl (9)
=H2(g) + Cl2 (9)
K = [H][CI]/[HCI)
UK = [H][CI]/2[HCI]
K = 2[HC]/[H]CI.)
K = [HCI)?/[H][1]
Answers
Answer:
(K) = [H2] [Cl2] / [HCl]²
Explanation:
The equation of the reaction is given as;
2 HCl (g) ⇔ H2 (g) + Cl2 (g)
The Equilibrium constant is the ratio of the concentration of the products and reactants raised to the power of their coefficients
Products = H2 and Cl2
Reactants = HCl
Equilibrium constant (K) = [H2] [Cl2] / [HCl]²
What should you do with a chemical burn?
A. use butter on the burn, bandage, and send the employee home to rest
B. use ice on the burn, bandage, and send the employee to the hospital
C.do not touch the victim, keep the victim quiet and call for medical assistance
D. call for medical assistance and check the MSDS for emergency interventions
Answers
Answer:
C. do not touch the victim keep the victim quiet and call dor medical assistance
Answer:
D.
Explanation:
MSDS if available should have tx instructions once a call for emergency medical assistance has been made
5. What is characteristic of an opaque object.